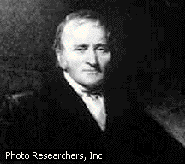Listed are pictures and short histories of those scientists who have contributed to the gas laws. Due to the purpose of this site, only a few of the scientists' discoveries will be discussed further on the following pages.
 Robert Boyle (1627-1691)
Robert Boyle (1627-1691)
Robert Boyle and his assistant Robert Hooke created an air pump which allowed for new experimentation concerning gases. In 1662 Boyle discovered the relationship between pressure and volume, now known (appropriately) as Boyle's law. At the time, no standard temperature scale existed, so he was unable to determine the relationship between the temperature and the volume, though he did understand that gases expanded when heated.
 Jacques Charles (1746-1823)
Jacques Charles (1746-1823)
Charles was a balloonist with very little formal training in math and science who experimented with the effect of temperature on the volume of gases. In 1783 he heard that someone had flown in a gas balloon. He assumed they had filled the balloon with hydrogen and soon began his own experiments. His work provided keys to understanding gases. Though he never published his findings, he is still credited for his work which has been summarized in Charles' law.
 Joseph Gay-Lussac (1778-1850)
Joseph Gay-Lussac (1778-1850)
Gay-Lussac is known for investigating the proportions of oxygen and hydrogen in water and other ratios of reacting gases. He found that if the product of the reaction of gases was a gas, it filled a volume directly related to the volume filled by the reacting gases. He also conducted experiments with the proportions of nitrogen and oxygen in the atmosphere, most notably ascending over 23,000 feet in a hot air balloon to measure oxygen high in the atmosphere.
 Amedeo Avagadro (1776-1856)
Amedeo Avagadro (1776-1856)
In 1811 Avagadro published an article stating that at a constant temperature and pressure, the same volumes of different gases contain exactly the same number of particles (6.022E23). He had based this idea on Gay-Lussac's discovery that all gases expand by the same amount when heated equally. His idea was largely ignored for nearly fifty years.
 John Dalton (1766-1844) ...or Mr. Sample???
John Dalton (1766-1844) ...or Mr. Sample???
Dalton was an English chemist and physicist who proposed that the pressure of a gas mixture is the sum of the individual gas pressures of each gas individually. This law is known as Dalton's law of partial pressures. He is also known for his work in meteorology and the development of the atomic theory.